
|

|
 |
 |

Parasite Primary Lit Papers
|
 |
 |
 |
 |
  I. I. | Parasite Papers Assignment Description
|
 |
 |
 |
 |
 |
  A] A] | In this exercise, you and a fellow student will read a paper from the scientific literature. All the papers in the exercise deal with changes wrought upon a host by a parasite. In each case, There will at minimum be a plausible argument as to how the changes could be favorable to the propagation of the parasite (most often by increasing the probability of the parasite ending up in its next host).
|
 |
 |
 |
 |
 |
  B] B] | You should pay attention to the structure of the paper itself since you will be performing your own research and writing it up formally during the course of the semester
|
 |
 |
 |
 |
 |
  1] 1] | Note that Appendix A of the lab manual explains the format and purpose of a general scientific paper
|
 |
 |
 |
 |
 |
  C] C] | In two or three weeks' time, you will hand in a brief single paragraph summary of the paper to your Lab Instructor, and present a summary of the paper to your classmates. The oral summary should include:
|
 |
 |
 |
 |
 |
  1] 1] | Description of host and parasite as organisms and where they appear on the Tree of Life
|
 |
 |
 |
 |
 |
  2] 2] | The change in host behavior/appearance wrought by the parasite, and how it could contribute to parasite propagation
|
 |
 |
 |
 |
 |
  3] 3] | A sketch on the whiteboard of a key figure from the paper (or one you synthesize from data in the paper) that demonstrates the influence of the parasite on the host
|
 |
 |
 |
 |
 |
  4] 4] | Presentation should be 5-7 minutes max; less is fine if you clearly convey the biology and data involved.
|
 |
 |
 |
 |
 |
  D] D] | You should arrive at class next week having found your paper and considered its presentation so you can discuss any questions with your Lab Instructor
|
 |
 |
 |
 |
 |
  E] E] | Note that the great majority of recent scientific literature is now available electronically; you can access such work at the University of Arizona from the following link (or use the Refences and Writing link from the course homepage):
|
 |
 |
 |
 |
 |
  2] 2] | Other journals can be found and xeroxed from the UA Science library
|
 |
 |
 |
 |
 |
  1] 1] | definitive host: the host in which the parasite performs sexual reproduction
|
 |
 |
 |
 |
 |
  2] 2] | intermediate host: a host in which the parasite does not undergo sexual reproduction
|
 |
 |
 |
 |
 |
  G] G] | Important note: you will be required to comment on/summarize the contents of your peers' presentations
|
 |
 |
 |
 |
 |
  A] A] | Infected amphipods show altered behavior and altered color; these alterations make infected amphipods more likely to be consumed by stickleback fish
|
 |
 |
 |
 |
 |
  1] 1] | Bakker T. C., M., D. Mazzi, and S. Zala 1997. Parasite-induced changes in behavior and color make Gammarus pulex more prone to fish predation. Ecology 78: 1098-1104.
|
 |
 |
 |
 |
 |
  B] B] | A parasite can alter the predator evasion behavior of an amphipod.
|
 |
 |
 |
 |
 |
  1] 1] | Bethel W. M. and J. C. Holmes 1973. Altered evasive behavior and responses to light in amphipods harboring acanthocephalan cystacanths. The Journal of Parasitology 59: 945-956.
|
 |
 |
 |
 |
 |
  C] C] | Parasite makes rats think cats are OK
|
 |
 |
 |
 |
 |
  1] 1] | Berdoy, M., J.P. Webster, D.W. Macdonald, 2000. Fatal attraction in rats infected with Toxoplasma gondii. Proceedings of the Royal Society B, 267(1452):1591-4.
|
 |
 |
 |
 |
 |
  D] D] | When stung in the head by a parasitoid wasp, cockroaches are stimulated to groom for extended periods of time.
|
 |
 |
 |
 |
 |
  1] 1] | Weisel-Eichler, A., G. Haspel, and F. Liberstat. 1991. Venom of a parasitoid wasp induced prolonged grooming in the cockroach. The Journal of Experimental Biology. 202:957-964.
|
 |
 |
 |
 |
 |
  E] E] | Parasite 'arranges' beetle's death pose to enhance further transmission
|
 |
 |
 |
 |
 |
  1] 1] | Carner, 1980 Entomophthora lampyridarum, a fungal pathogen of the soldier beetle, Chauliognathus pennsylvanicus Journal of Invertebrate Pathology, 36: 394-398
|
 |
 |
 |
 |
 |
  2] 2] | Data = images & measurements
|
 |
 |
 |
 |
 |
  F] F] | Two parasites with different definitive hosts infect the same host in an amphipod-acanthocphalan system.
|
 |
 |
 |
 |
 |
  1] 1] | Cezilly F., A. Gregoire, and Bertin A. 2000. Conflict between co-ocurring manipulative parasites? An experimental study of the joint influence of two acanthocephalan parasites on the behavior of Gammarus pulex. Parasitology 120: 625-630.
|
 |
 |
 |
 |
 |
  G] G] | Wasp parasite induces spider to spin web useful to parasite as coccoon
|
 |
 |
 |
 |
 |
  1] 1] | Eberhard, W. G. 2001. Under the influence: webs and building behavior of Plesiometa argyra (Araneae, Tetragnathidae) when parasitized by Hymenoepimecis argyraphaga (Hymenoptera, Ichneumonidae). Journal of Arachnology 29:354-66.
|
 |
 |
 |
 |
 |
  H] H] | Altered behavior of isopod host is initiated by the parasite in order to facilitate transmission to a fish definitive host.
|
 |
 |
 |
 |
 |
  1] 1] | Hechtel L. J., C. L. Johnson, and S. A. Juliano 1993. Modification of antipredator behavior of Caecidotea Intermedius by its Parasite Acanthocephalus Dirus. Ecology 74: 710-713.
|
 |
 |
 |
 |
 |
  I] I] | Copepods that are infected by a parasite show altered response to the odor stimuli of fish predators. Results in increased predation by fish definitive host.
|
 |
 |
 |
 |
 |
  1] 1] | Jakobsen P. J. and C. Wedekind 1998. Copepod reaction to odor stimuli influenced by cestode infection. Behavioral Ecology 9: 414-418.
|
 |
 |
 |
 |
 |
  J] J] | Infection by the trematode Crassiphiala bulboglossa causes the banded killifish to engage in higher predation-risk behavior.
|
 |
 |
 |
 |
 |
  1] 1] | Krause, J. and J.-G. Godin,1994 Influence of parasitism on the shoaling behaviour of banded killifish, Fundulus diaphanus. Canadian Journal of Zoology, 72:1775
|
 |
 |
 |
 |
 |
  K] K] | Parasites cause infected killifish to alter their behavior. The altered behavior results in increased predation of killifish by final avian hosts of the parasite.
|
 |
 |
 |
 |
 |
  1] 1] | Lafferty K. D. and A. K. Morris 1995. Altered behavior of parasitized killifish increases susceptibility to predation by bird final hosts. Ecology 77: 1390-1397.
|
 |
 |
 |
 |
 |
  L] L] | Magnitude of parasitization influences growth rate and risk-avoidance behavior of Macoma balthica.
|
 |
 |
 |
 |
 |
  1] 1] | Lim, S.S.L. and R. H. Green, The relationship between parasite load, crawling behaviour, and growth rate of Macoma balthica (L.) (Mollusca, Pelecypoda) from Hudson Bay, Canada 1991 Canadian Journal of Zoology, 69:2202
|
 |
 |
 |
 |
 |
  M] M] | Phenotype of threespine stickleback is altered by parasitization with Schistocephalus solidus.
|
 |
 |
 |
 |
 |
  1] 1] | LoBue, C.P. and M.A. Bell, Phenotypic Manipulation by the Cestode Parasite Schistocephalus solidus of Its Intermediate Host, Gasterosteus aculeatus, the Threespine Stickleback, 1993 The American Naturalist, 142:725
|
 |
 |
 |
 |
 |
  2] 2] | data is integrated in text
|
 |
 |
 |
 |
 |
  N] N] | An acanthocephalan parasite affects amphipods by making them more likely to be found drifting in the water column.
|
 |
 |
 |
 |
 |
  1] 1] | Maynard B. J., T. A. Wellnitz, N. Zanini, W. G. Wright, and B. S. Dezfuli 1998. Parasite-Altered Behavior in a Crustacean Intermediate Host: Field and Laboratory Studies. Journal of Parasitology 84: 1102-1106.
|
 |
 |
 |
 |
 |
  O] O] | A fungus kills its fly host. Dead flies have distended abdomens which cause male flies to approach dead flies and attempt to mate with them.
|
 |
 |
 |
 |
 |
  1] 1] | Moller, A. P. 1993. A fungus infecting domestic flies manipulates sexual behaviour of its host. Behavioural Ecology and Sociobiology. 33:403-407.
|
 |
 |
 |
 |
 |
  P] P] | Infected cockroaches show altered behavior as compared to uninfected conspecifics.
|
 |
 |
 |
 |
 |
  1] 1] | Moore J. 1983a. Altered behavior in Cockroaches (Periplaneta americana) infected with an Archiacanthocephalan, Moniliformis moniliformis. Journal of Parasitology 69: 1174-1176.
|
 |
 |
 |
 |
 |
  Q] Q] | Isopods that are infected by an acanthocephalan parasite show altered behavior and are more likely to be consumed by avian predators that serve as the definitive host.
|
 |
 |
 |
 |
 |
  1] 1] | Moore J. 1983b. Responses of an avian predator and its isopod prey to an acanthocephalan parasite. Ecology 64: 1000-1015.
|
 |
 |
 |
 |
 |
  R] R] | Infected cockles are unable to bury themselves in the mud, and are thus more likely to be predated by definitive hosts.
|
 |
 |
 |
 |
 |
  1] 1] | Mouritsen K. N. 2002. The parasite-induced surfacing behaviour in the cockle Austrovenus stutchburyi: a test of an alternative hypothesis and identification of potential mechanisms. Parasitology 124: 521-528.
|
 |
 |
 |
 |
 |
  S] S] | Young female mice infected with Spirometra mansonoides show accelerated weight gain, even to the point of obesity.
|
 |
 |
 |
 |
 |
  1] 1] | Mueller, J.F. 1963 Parasite induced weight-gain in mice. Annals of the New York Academy of Sciences, 113:217
|
 |
 |
 |
 |
 |
  T] T] | 'hairworms' drive their terrestrial insect hosts to enter water
|
 |
 |
 |
 |
 |
  1] 1] | Thomas et al. Do hairworms (Nematomorpha) manipulate the water seeking behaviour of their terrestrial hosts? 2002 Journal of Evolutionary Biology 15:356-61
|
 |
 |
 |
 |
 |
  U] U] | Threespine stickleback infected with the tapeworm Schistocephalus solidus behave in a manner which increases their exposure to piscivorous birds, the definitive host of S. solidus
|
 |
 |
 |
 |
 |
  1] 1] | Ness, J.H., S.A. Foster 1999. Parasite-associated phenotype modifications in threespine stickleback. Oikos 85:127
|
 |
 |
 |
 |
 |
  V] V] | Acanthocephalan infections cause some isopods to change color and/or size
|
 |
 |
 |
 |
 |
  1] 1] | Oetinger, D.F. and B. B. Nichol 1981 Effects of acanthocephalans on pigmentation of freshwater isopods. Journal of Parasitology, 67:672
|
 |
 |
 |
 |
 |
  W] W] | A tapeworm is documented that produces a host growth hormone. It is speculated that the hormone could affect the host and result in increased predation by the definitive host of the parasite.
|
 |
 |
 |
 |
 |
  1] 1] | Phares, K. 1996. An unusual host-parasitoid relationship: the growth hormone-like factor from Plerocerdoids of Spirometrid Tapeworms. International Journal for Parasitology. 26: 575-588.
|
 |
 |
 |
 |
 |
  X] X] | Mosquitoes were able to feed more efficiently on infected hosts, leading to increased transmission of the pathogen that causes malaria.
|
 |
 |
 |
 |
 |
  1] 1] | Rossignol P.A., Ribeiro J.M.C., Jungery M., Turell M.J., Spielman A. & Bailey C.L. 1985. Enhanced Mosquito Blood-Finding Success on Parasitemic Hosts - Evidence for Vector Parasite Mutualism. Proceedings of the National Academy of Sciences of the United States of America 82: 7725-7727.
|
 |
 |
 |
 |
 |
  Y] Y] | Parasitism alters bumblebee flower pollination preference.
|
 |
 |
 |
 |
 |
  1] 1] | Schmid-Hempel, P. and S. Hans-Peter. 1998. Parasites and flower choice of bumblebees. Animal Behaviour. 55: 819-825.
|
 |
 |
 |
 |
 |
  Z] Z] | Parasitoid causes host caterpillar defend parasite young
|
 |
 |
 |
 |
 |
  1] 1] | Grosman, Amir H., A. Janssen, E. F. de Brito, E. G. Cordeiro, F Colares, J. Oliveira Fonseca, E. R. Lima, A. Pallina, M. W. Sabelis. Parasitoid increases survival of its pupae by inducing hosts to fight predators. PLoS ONE 3(6): e2276 doi:10.1371/journal.pone.0002276
|
 |
 |
 |
 |
 |
  2] 2] | Note: PLoS ONE is an on-line, peer-reviewed journal. You can search for this article by going to www.plosone.org
|
 |
 |
 |
 |
 |
  AA] AA] | [Not electronically available; students welcome to hunt hardcopies]
|
 |
 |
 |
 |
 |
  1] 1] | Infection of brown and black cockroaches by Moniliformis moniliformis results in different behavioral changes for the two types of cockroach
|
 |
 |
 |
 |
 |
  a. a. | Carmichael, 1990 Journal of Parasitology, 77:931
|
 |
 |
 |
 |
 |
  2] 2] | Isopods parasitized by Acanthocephalus jacksoni behave in a way that may make them more conspicuous to predators
|
 |
 |
 |
 |
 |
  a. a. | Muzzall,1962 Proceedings of the Helmithological Society of Washington 42:116
|
 |
 |
 |
 |
 |
  3] 3] | Guppies parasitized by Diplostomum spathaceum are more suseptible to predation
|
 |
 |
 |
 |
 |
  a. a. | Brassard, 1982 Parasitology, 85:495
|
 |
 |
 |
 |
 |
  III. III. | Leads for future material
|
 |
 |
 |
 |
 |
  1] 1] |
Copyright Pontoppidan et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Graveyards on the Move: The Spatio-Temporal Distribution of Dead Ophiocordyceps-Infected Ants
Maj-Britt Pontoppidan,1* Winanda Himaman,2 Nigel L. Hywel-Jones,3 Jacobus J. Boomsma,4 and David P. Hughes4,5,6*
|
 |
 |
 |
 |
 |
  a. a. | Thorny-headed worms begin their life cycle inside invertebrates that reside in marine or freshwater systems. Gammarus lacustris, a small crustacean that feeds near ponds and rivers, is one invertebrate that the thorny-headed worm may occupy. This crustacean is preyed on by ducks and hides by avoiding light and staying away from the surface. However, when infected by a thorny-headed worm it becomes attracted toward light and swims to the surface. Gammarus lacustris will even go so far as to find a rock or a plant on the surface, clamp its mouth down, and latch on, making it easy prey for the duck. The duck is the definitive host for the acanthocephalan parasite. In order to be transmitted to the duck, the parasite's intermediate host (the gammarid) must be eaten by the duck. This modification of gammarid behavior by the acanthocephalan is thought to increase the rate of transmission of the parasite to its next host by increasing the susceptibility of the gammarid to predation.
It is thought that when Gammarus lacustris is infected with a thorny-headed worm, the parasite causes serotonin to be massively expressed. Serotonin is a neurotransmitter involved in emotions and mood. Researchers have found that during mating Gammarus lacustris expresses high levels of serotonin. Also during mating, the male Gammarus lacustris clamps down on the female and holds on for days. Researchers have additionally found that blocking serotonin releases clamping. Another experiment found that serotonin also reduces the photophobic behavior in Gammarus lacustris. Thus, it is thought that the thorny-headed worm physiologically changes the behavior of the Gammarus lacustris in order to enter its final host, the bird.
|
 |
 |
 |
 |
 |
  a. a. | The investigation of a rampaging cricket STD that makes its victims both sterile and rabidly lustful led a Dalhousie University researcher to a somewhat less salacious discovery that could, with further exploration, have meaningful implications for human treatment options.
Biologist Dr. Shelley Adamo published a research paper titled, “A viral aphrodisiac” in the Journal of Experimental Biology last month.
She said her colony of crickets contracted the highly virulent IIV-6/CrIV virus from a reptile brought into the lab for a separate study.
She said she noticed her females had stopped laying eggs, though they seemed otherwise completely healthy – not lethargic and eating normally.
“When I opened up a female, I was shocked. This female was not a little sick, she was a lot sick,” said Adamo Thursday, adding the cricket had bloated, bluish fat cells where her eggs should have been.
After confirming the presence of the virus, Adamo said she determined that the virus was responsible for significantly increasing their sexual appetites – an effect not documented in any cricket pathogen.
However, what’s more notable than the aphrodisiac effect is that the virus severed communication between the immune and central nervous systems, blocking normal ‘sick’ behaviours such as lethargy or reduced appetite.
“My guess is, it does what viruses do, which is hijack the genome of its host,” she said. “It’s forcing the host to make viral proteins and suppressing the expression of immune-related proteins. And the actual molecular details of that would be cool to figure out, but we didn’t get that far.”
Adamo said sexually transmitted infections in most mammals are also asymptomatic, suggesting a similar “blocking” mechanism that’s beneficial to the continued survival of the disease.
She said figuring out how viruses suppress immune signals could lead to insights in regulating the intracellular signaling systems – which could have important implications for some human treatment therapies that involve stimulating the immune system.
“Unfortunately, they have serious side effects, and most of these side effects have to do with overactivating these communication signals, so people feel extremely ill,” she said. “So…there would be benefits if you could figure out how to regulate that without suppressing immune function, that would be the key.”
|
 |
 |
 |
 |
 |
  1] 1] | Meet the Wasp that Turns Spiders into Zombie Construction Workers
Matt Soniak
In another post, we were talking about what happens to spiders and their webs when scientists give them a little bit of marijuana and other drugs. While researching that post, I learned that its not just humans that alter spiders’ behavior with chemicals. There’s a wasp in Costa Rica that does the same thing, in a more gruesome and sinister way, as part of its journey to adulthood.
The tropical wasp species Hymenoepimecis argyraphaga is a parasite, and it takes advantage of an unlikely host. The web of the orb weaver spider Plesiometa argyra is normally a place where bugs meet their untimely death and become spider snacks. Using an arsenal of toxins and mind-altering chemicals, though, H. argyraphaga is able to turn the spider into a slave and a meal and its web into a safe haven.
When the female wasp is ready to lay her egg, she seeks out a spider to help raise her child. She stings the spider to paralyze it and then lays an egg on its abdomen. Soon, the egg hatches and the larva that emerges remains attached to the spider, living on its abdomen and sucking hemolymph (kind of the arthropod version of blood) from its body for nourishment. After a few weeks like this, during which the spider goes about its life as normal, the wasp larva is ready to move on to the next stage of its life cycle. To do this, it needs to make a cocoon. A spider’s web is a decent place to build one, but not perfect. It’s suspended above the ground and the sticky threads provide defense from predators that might eat the wasp, but it’s far too flimsy to support the heavy cocoon and the adult wasp that will come out of it.
The wasp gets around this problem by forcing the spider to build a web that suits its needs. It injects a chemical (which hasn’t been identified yet) into the spider that alter’s the host’s behavior. The spider begins building it’s web as normal, but instead of completing all the steps and getting a carefully-patterned web, the zombified spider simply repeats the first few steps over and over again, resulting in a web that’s just a few heavily-reinforced anchor threads and a small center section.
Once the web is done, the spider crawls to the center of the web and sits there complacently. The larva molts, kills the only companion it has ever known, sucks any remaining useful bits out from its corpse, and discards it. Then, it builds its cocoon on a web custom-built for the job. A few weeks later, the adult wasp emerges and flies away, and the cycle starts over.
For more on parasitic wasps, see any of Carl Zimmer’s paeans to the Emerald Cockroach Wasp.
|
 |
 |
 |
 |
 |
  a. a. | A spiny ant, Polyrhachis armata, from Thailand biting a rainforest leaf following infection by the fungus Ophiocordyceps unilateralis. The large stalk and two spore-producing bodies are visible. Image and caption courtesy David Hughes.
The zombie is a simple creature with simple tastes, enjoying leisurely walks on the beach, dining out with hordes of its friends, and every now and then having a good tumble down a flight of stairs. It behaves this way because the pathogen that has infected it doesn’t require complex behaviors in order to replicate — it commands a hungry, nearly indestructible vessel that can walk it right up to its next potential host.
But on our planet there exist zombified ants that undergo a decidedly more complex, and more disturbing, transformation at the hands of highly sophisticated parasitic fungi that assume control of the insects’ minds. What ensues between a host and a parasite with no brain of its own is a battle that is far stranger and far more methodical than anything ever dreamed up by Hollywood. (The zombifying fungus that attacks humans in the videogame The Last of Us comes close, but its real-life counterpart is much, much weirder. And you don’t have to pay 60 bucks to see it, which is nice.)
For many of us it’s hard to feel for ants, what with them ruining picnics or even entire cities, but it’s downright disquieting to watch one infected by these parasitic fungi — species in the genus Ophiocordyceps that each, incredibly, attack only a single species of ant. Once a disciplined member of a rigidly structured society, the affected ant stumbles out of its colony like the town drunkard, guided by a pathogen that has pickled its brain with a cocktail of chemicals.
Once a disciplined member of a rigidly structured society, the affected ant stumbles out of its colony like the town drunkard, guided by a pathogen that has pickled its brain with a cocktail of chemicals.
The ant heads, at the behest of the fungus, to a precise position in the forest. Scientists plotting the coordinates of these unfortunate ants have documented a striking regularity to their travels, making the pathogen a bit like GPS for the insect, only, you know, the ant never asked for directions.
The ants “are manipulated to bite onto very specific locations on the underside of a leaf, the main vein of a leaf, leaves orientated north, northwest, roughly 25 cm off the ground,” said David Hughes, a behavioral ecologist at Penn State. “And all of this happens with a remarkable precision around solar noon, making this one of the most complex examples of parasite manipulation of host behavior.”
It’s a position chosen by the fungus, rather unbelievably, for its ideal temperature and humidity — Hughes has experimented with this by moving the ants out of these spots to drier, hotter areas, where the fungus failed to grow. Once the ant has anchored itself by sinking its mandibles into the leaf’s vein, it perishes, and from the back of its head erupts a stalk, which, while in a way is quite beautiful, might be considered the world’s least desirable hat. This in turn rains spores down onto the ant’s fellow workers below, attaching to their exoskeletons and beginning what could euphemistically be called an invasive procedure.
“In order to get through [the exoskeleton], the fungus builds up a pressure,” said Hughes. “We know from studies of fungal parasites of plants, particularly rice, they can build up a pressure inside their spore equivalent to the pressure in the wheel of a 747. So they have a massive buildup of pressure, and when that’s at a sufficient level then they blow a hole through the wall and blow all the genetic material” into the ant.
Thus the cycle begins anew.
According to Hughes, in addition to the 160 known species of ant-controlling fungi, there may be some 1,000 additional varieties out there to be discovered. These don’t even account for the array of additional parasitic fungi that exclusively target specific species of other insects, from beetles to butterflies (let’s face it, butterflies could use to get taken down a notch or two).
The relationship is a remarkable illustration of host-parasite coevolution that scientists are just beginning to understand — fossil records of bite-scarred leaves show this has been happening (.pdf) for at least 48 million years — with ant-hunters, each dependent on a single species, developing astounding adaptations to survive. And in response, the ants have evolved their own brilliant defenses, far beyond anything you learned from SimAnt.
“The fungus needs to transmit,” said Hughes, “and it cannot do that inside the nest, because in order for ant societies to work, they have necessarily evolved a prophylactic immune system, which is reliant upon behavioral defense. So they have something called social immunity. They simply stop diseases spreading inside their nest by finding diseased individuals and moving them out.”
Despite the ants’ countermeasures, these fungi are extremely virulent and can, as if trying to show off, wipe out whole colonies. Left unchecked, the fungi might conceivably drive themselves and their ant hosts to extinction. But this is where the tale gets stranger. The parasitic fungi themselves have their own parasitic fungi.
The very success that allows the fungi to build up what Hughes calls “graveyards in the forest” also “invites other organisms to come in and infect them,” he said. “And these hyperparasitic fungi castrate the zombie ant fungi. So the zombie ant fungi rely upon a spore body that releases spores and continues to cycle, and the other parasite comes in and whacks it out.” In one study Hughes found that only 6.5 percent of a zombie ant fungi’s fruiting bodies produced viable spores.
The whole weird circus is still somewhat mysterious, but Hughes is studying infected ants in the lab to figure out what kinds of chemicals the fungi are using to achieve mind-control, and how exactly mind-control affects transmission. These species, after all, are not alone among fungi in their psychoactive tendencies. LSD was synthesized from ergot, a rye-loving fungus theorized, though far from proven, to have tripped out the poor souls accused in the Salem Witch Trials, which it turns out wasn’t nearly as groovy of a situation as it sounds, on account of all the capital punishment.
“We’re discovering that over half of life on Earth is parasitic,” Hughes said. “It’s the most common mode of existence in the history of life on Earth. But only a tiny minority of parasites do mind-control. And why is that? What is the push in order to control the behavior of your host?”
Other than to enjoy a leisurely stroll on the beach, of course.
Further reading: Those interested in academic explorations of zombie fungi and other pathogens can sign up for Penn State’s free online course Epidemics: The Dynamics of Infectious Diseases, which begins Oct. 15.
|
 |
 |
 |
 |
 |
  2] 2] | There is much evidence that some pathogens manipulate the behaviour of their mosquito hosts to enhance pathogen transmission. However, it is unknown whether this phenomenon exists in the interaction of Anopheles gambiae sensu stricto with the malaria parasite, Plasmodium falciparum - one of the most important interactions in the context of humanity, with malaria causing over 200 million human cases and over 770 thousand deaths each year. Here we demonstrate, for the first time, that infection with P. falciparum causes alterations in behavioural responses to host-derived olfactory stimuli in host-seeking female An. gambiae s.s. mosquitoes. In behavioural experiments we showed that P. falciparum-infected An. gambiae mosquitoes were significantly more attracted to human odors than uninfected mosquitoes. Both P. falciparum-infected and uninfected mosquitoes landed significantly more on a substrate emanating human skin odor compared to a clean substrate. However, significantly more infected mosquitoes landed and probed on a substrate emanating human skin odor than uninfected mosquitoes. This is the first demonstration of a change of An. gambiae behaviour in response to olfactory stimuli caused by infection with P. falciparum. The results of our study provide vital information that could be used to provide better predictions of how malaria is transmitted from human being to human being by An. gambiae s.s. females. Additionally, it highlights the urgent need to investigate this interaction further to determine the olfactory mechanisms that underlie the differential behavioural responses. In doing so, new attractive compounds could be identified which could be used to develop improved mosquito traps for surveillance or trapping programmes that may even specifically target P. falciparum-infected An. gambiae s.s. females.
|
 |
 |
 |
 |
 |
  H] H] | Parasite review papers
|
 |
 |
 |
 |
 |
  I] I] | Review article (popular)
|
 |
 |
 |
 |
 |
  a. a. | Heads up. I'm starting with a spoiler. One of the most effectively scary movie moments I’ve ever seen is in the otherwise unmemorable 1979 When A Stranger Calls. A young babysitter keeps getting phone calls asking “have you checked the children?” Eventually she tires of the prank and contacts the police. They tell her to make sure all the doors are locked and if it happens again to keep the stranger on the line. The next time he rings she manages to get a few more words out of him, but he still hangs up quickly. The police call back. "We’ve traced the call. It’s coming from inside the house”.
When I first heard that line some screamed, some jumped, and I yelled because my girlfriend at the time nearly pulled my arm off. What makes it so much more unnerving and disturbing than all the typical horror fare which follows is exactly what this month’s bidding blockbuster, The Host, has become the latest film to tap into. The enemy within.
Just like The Puppet Masters, David Cronenberg’s Shivers and many mostly B-movies, The Host centres on that sci-fi staple of an occupying organism which crawls onto - or more usually into – humans, taking near total control of them. But rather than this leading to a horrible transformation which is revealed, reviled and resisted over the course of the film, from the first frames of The Host we are in a future where the takeover is already almost complete. And where Earth seems all the better for it.
This makes it a neat variant both on invading life forms who want to eat us, or enslave us or just eliminate us, and on the cuddly ETs who just want to be our friends. Here the alien parasites, known as souls, have made the world a more joyous and harmonious place through the simple expedient of replacing our own natures with theirs, which by contrast are “compassionate, patient, honest, virtuous and full of love”. Wonderful, serene and wise as these souls (mostly) are, they only prosper by suppressing or snuffing out all those facets and flaws which makes each of us human. It’s smiley genocide.
Mind benders
If the idea of our species being enhanced by parasitic “souls” is ringing a distant bell, it might be that you’re reminded of Brian Aldiss’ 1962 novel Hothouse in which sentient fungi called morels boost our intelligence by attaching themselves to our heads. Either that or the many real world examples of parasitic behaviour that seem like they come straight from the movies.
There are viruses, flukes, wasps, tapeworms, fungi and a whole host of tiny creatures whose only goal in life is to infect, exploit and take control of their host’s every day behaviour.
Take grasshoppers infected with hairworm. One day they are going about their business; the next they find themselves compelled to jump into pools of water. The parasite essentially makes grasshoppers suicidal to ensure they can reproduce.
Then there is the gammarid – a tiny shrimp-like crustacean that are a favourite of the thorny-headed worm. Your normal, uninfected gammarid is a shy creature that when threatened with being eaten, will dive into the mud at the bottom of the pond they live. But once infected, the creature does the opposite – shooting towards the surface. Again, the host has taken control of its senses – turning the gammarid into a zombie and allowing it to complete its life cycle by being eaten by a bird.
Or there is the Lancet liver fluke, an ingenious creature that goes from cow, to snail to ant and back again – the really clever bit coming at the end of the life cycle where the mind bending creature forces the unwitting ants to climb to the top of a blade of grass night after night, until it is eaten by a cow, starting the whole cycle again.
Love drug
And behaviour bending may also happen closer to home. Several studies have shown over the years that we too may be under the influence. With a stress on the “may”.
It has been known for a long time that rats infected with the single-celled brain parasite Toxoplasma gondii – commonly found everywhere form soil, infected meat and cat faeces – causes them to lose their fear. Stick an infected rat in a room with a cat and it will carry on oblivious to its inevitable fate.
And humans - of which a third may be infected with Toxoplasma gondii - could have similar tendencies (to lose their fear, rather than to sit happily in a room with a cat). A 2006 study, for example, suggested the infection could cause subtle but significant personality changes – potentially affecting everything from guilt, intelligence and affection. The author even raises the question of whether the creature has influenced human culture.
Which brings us back to The Host. There is a bit more to the film than just parasitic performance, and – being based on a novel by Stephenie “Twilight” Meyer – there’s also room for oodles of complicated romantic geometry way beyond bizarre love triangles, with people and their souls falling collectively or separately for other people and/or their souls. I said it was complicated.
Had The Host been made a few decades ago, it might have been taken as a thinly veiled parable about the threat of communism: a poor cousin to that greatest of alien invasion movies, the original 1956 Invasion of the Bodysnatchers (which, for the record, all those involved with have always insisted was just a thriller, nothing more). Nowadays, although Meyer herself claims a key theme is learning to love the bodies we are in, some have viewed The Host as echoing contemporary concerns about the over use of drugs that may calm us down or cheer us up, but subdue our personalities as well as our anxieties. Others see it as being about mental disease and decay, and the fear that our self slips away from our body. And that’s all before – at the time of writing – the film has even been released.
Even in the hands of Andrew Niccol, the writer/director responsible for such thought-provoking science fiction as Gattaca and The Truman Show (and more recently but less effectively In Time) I’m not sure that The Host is able to bear the weight of all those possibilities. It is mostly just a teen-friendly sci-fi romance with a love conquers all message. But perhaps all these interpretations and extrapolations reveal another parasitic relationship, the one in which different audiences burrow into movies and find what they need to feed off within them.
|
 |
 |
 |
 |
 |
  2] 2] | Four new Brazilian species in the genus Ophiocordyceps have been published in the online journal PLoS ONE. The fungi, named by Dr Harry Evans and Dr David Hughes, belong to a group of 'zombifying' fungi that infect ants and then manipulate their behaviour, eventually killing the ants after securing a prime location for spore dispersal.
These results appear in a paper by Evans et al. entitled Hidden Diversity Behind the Zombie-Ant Fungus Ophiocordyceps unilateralis: Four New Species Described from Carpenter Ants in Minas Gerais, Brazil. This paper is the first to validly publish new fungal names in an online-only journal while still complying with the rules and recommendations of the International Code of Botanical Nomenclature (ICBN).
Beyond this important milestone, the paper is noteworthy for the attention it draws to undiscovered, complex, biological interactions in threatened habitats. The four new species all come from the Atlantic Rainforest of Brazil which is the most heavily degraded biodiversity hotspot on the planet. Ninety-two percent of its original coverage is gone.
The effect of biodiversity loss on community structure is well known. What researchers don't know is how parasites, such as these zombie-inducing fungi, cope with fragmentation. Here the authors show that each of the four species is highly specialised on one ant species and has a suite of adaptations and spore types to ensure infection. The life-cycle of these fungi that infect, manipulate and kill ants before growing spore producing stalks from their heads is remarkably complicated. The present work establishes the identification tools to move forward and ask how forest fragmentation affects such disease dynamics.
|
 |
 |
 |
 |
 |
  K] K] | PLANT behavior changer
|
 |
 |
 |
 |
 |
  a. a. | Fungus, Get Off My Lawn!
by Elizabeth Pennisi on 1 March 2013, 4:00 PM |
ENLARGE IMAGE

Greedy guest. Fungi (blue lines) living among the cells of a grass cause that plant to make more seeds and less pollen.
Credit: Jennifer Rudgers
Life demands tradeoffs, and plants are no exception. Virginia wildrye, common on U.S. prairies and rangelands, often plays host to a fungus that helps this grass grow. But the plant pays a price. Researchers have discovered that infected plants produce less pollen than their noninfected counterparts. Instead, the fungus causes the rye grass to make extra seeds, which transmit the fungus to the next generation and new locations. This is the first time a fungus has proven capable of manipulating plant reproduction. The finding highlights the complexity of the relationship between hosts and their guests.
Relationships abound in which a plant or animal partners with another organism. That organism, called a symbiont, gets protection from the environment and, in return, sometimes offers some benefit. In the case of Virginia wildrye, the microscopic fungus thrives between the cells of the stem and leaves. It makes a toxin that deters insects and other animals from eating the plant and also protects its host against disease and drought.
But Jennifer Rudgers, a botanist at the University of New Mexico, Albuquerque, who led the new study, began to wonder if there was more to the relationship. Even though Virginia wildrye is hermaphroditic, producing seeds (the female contribution) and pollen (male contribution), the fungus gets passed on to the next generation only in seeds. Pollen is too small to carry the fungus, she notes. "Basic theory predicts that whenever a symbiont is preferentially transmitted through one sex, it will evolve [ways] to overproduce that sex," explains John Werren, an evolutionary geneticist at the University of Rochester in New York who was not involved with the work. The same thing might hold true for an organism's sex cells, such as pollen and eggs. Many insects, for example, host microbes that are "inherited" through the eggs but not through sperm. The microbes often cause more female insects to be produced. They may convert genetic males into females, allow females to produce only daughters without the need for mating, or kill male offspring of infected females so that the daughters have more food. "These 'strategies' enhance transmission of the microbe," Werren says.
Rudgers decided to test whether the fungus was skewing the grass's reproductive efforts in a similar way. She compared pollen and seed production in plants that were infected with the fungus and plants that were not.
Infected plants produced one-quarter more seeds than noninfected plants, she and her colleagues have reported in the April issue of The American Naturalist. But the tradeoff was that they produced only about half the usual amount of pollen. Because Virginia wildrye is wind-pollinated, much of the pollen fails to reach its target and thus quite a lot is needed to assure adequate pollination occurs, Rudgers says. "They are not able to reproduce as they usually would," she notes. It is not clear whether the increase in seed number can make up for there being less pollen.
"There is a fairly dramatic effect on that tradeoff," says Christopher Schardl, a biologist at the University of Kentucky, Lexington, who was not involved with the work. "There's clearly an advantage to the fungus."
"To my knowledge, it is the first clear demonstration that symbionts can alter the seed to pollen ratio in plants," Werren says.
Schardl has some evidence to support this finding. He has been studying differences in gene expression in infected and uninfected plants and finds that the fungus seems to increase the activity of genes involved in seed production. Given that fungal symbionts are quite common in some plants, "it would be interesting to know how general this conclusion is," he notes.
|
 |
 |
 |
 |
 |
  1] 1] | Taking Aim
Photograph by Kees van der Krieke, Stippen.nl
The parasitic wasp Dinocampus coccinellae prepares to inject a spotted ladybug with a single egg in a file picture. The ladybug has been paralyzed by the wasp's venom.
In time the egg will hatch into a larva that will develop for a few days and then chew a small hole through the abdomen of the ladybug. The larva will then spin a cocoon between the legs of the ladybug, whose body will rest on top of the cocoon as the larva undergoes metamorphosis. (See insect-egg pictures.)
In a recent study in the journal Biology Letters, scientists note that sometimes the ladybugs survive the larva's emergence, and in those cases, the D. coccinellae larva then "brainwashes" the bug into defending the vulnerable cocoon from predators, said study co-author Jacques Brodeur, a biologist at the University of Montreal.
"The parasite is taking control of the behavior of its host—that's why we call it bodyguard manipulation," said Brodeur, who worked with Ph.D student Fanny Maure.
(See "'Zombie' Roaches Lose Free Will Due to Wasp Venom.")
|
 |
 |
 |
 |
 |
  2] 2] | Attacks of the Brain-Controlling Parasites
Once upon a time, parasites were thought to live relatively simple lives: They hitched a ride on a host, sapping nutrients and energy but otherwise leaving it alone. But that was only part of the story. Many parasites actually take control, causing their hosts to act in self-destructive ways that further their invaders' interests.
The Lymantria dispar baculovirus, for example, causes caterpillars to climb into treetops rather than hiding in bark. When those that go uneaten by birds finally die and decompose (as pictured above), viral particles rain onto foliage below, infecting a new generation of caterpillars.
"I think the reason people are a little creeped out by seeing pathogens control behavior is that we have examples of it around us all the time," said chemical ecologist Kelli Hoover of Pennsylvania State University, who describes L. dispar's gene target in a Sept. 9 Science study.
The following pages show more examples of parasites that spread by controlling their hosts.
|
 |
 |
 |
 |
 |
  2] 2] | Zombie ants: When ants become walking dead

A parasitized ant. Image credit: David P. Hughes. |
|
|
An ingeniously deadly fungus hijacks the bodies of ants for food and reproduction.
|
|
DOWNLOAD❘EMBED It’s like something out of a horror movie. A parasitic fungus infiltrates the body of a tropical carpenter ant, feeding on it and manipulating its body. The fungus forces the dying ant to the forest understory, an environment more conducive to its growth. The invasion of this fungal body-snatcher culminates with it sprouting a spore-laden fruiting body from the dead ant’s head.
An account of this deadly assault on tropical carpenter ants (Camponotus leonardi) by a parasitic fungus (Ophiocordyceps unilateralis) is described in the May 9, 2011 issue of the open access journal BMC Ecology.
Similar incidents of fungi parasitizing insects occur in other parts of the world. This particular case of zombie ants plays out in the forests of Thailand.
It’s a pretty gruesome affair, so if you find movies like The Thing and Night of the Living Dead too scary, now’s a good time to stop reading!
The paper’s lead author, Dr. David Hughes of Penn State University, described the ant-fungus interaction in a press release.
The fungus attacks the ants on two fronts. Firstly by using the ant as a walking food source, and secondly by damaging muscle and the ant’s central nervous system, resulting in zombie walking and the death bite, which place the ant in the cool damp understory. Together these provide the perfect environment for fungal growth and reproduction. This behavior of infected ants is essentially an extended phenotype of the fungus (fungal behaviour through the ant’s body) as non-infected ants never behave in this way.

A dead carpenter ant attached to a leaf in the understory of a forest in Thailand. Before killing the ant, the fungus growing in the ant changed its behavior, causing it to bite into the leaf vein. Image Credit: David Hughes, Penn State University.
Tropical carpenter ants spend most of their time high in the forest canopy. When they venture down to the jungle understory, they follow well-defined trails. It’s during this time that ants could get infected by fungal spores that land on their outer body.
The fungus can only complete its life cycle through the ant. Spores germinate, and the fungus penetrates the ant’s body. It proceeds to infect the entire animal, affecting its central nervous system. You can tell when a carpenter ant has been infected: instead of marching purposefully down a trail, an infected worker ant walks about haphazardly, displaying erratic behavior. Sporadic convulsions set in, causing the infected ant to fall from the canopy to the moist, cool, leafy forest understory, ideal conditions for the fungus to continue its growth.

A carpenter ant attached to a leaf. The ant has been dead for two to three days, and the fungus's fruiting body filled with spores sticks out of its head. Image Credit: David Hughes, Penn State University.
Infected ants on the forest understory are driven by the fungus to select leaves of saplings that are about 25 centimeters (10 inches) above the soil surface. Then, a curious thing happens when the sun shines at its highest intensity of the day, at solar noon when it reaches the highest point in the sky. The fungus commands the ant to sink its mandibles into the leaf’s main vein, on the underside of the leaf. A possible reason for this action is to attach the ant to a stable environment suitable for the fungus’s subsequent development. But this synchronization with solar noon is a mystery, and it will be the subject of follow-up research.
Scientists call this stage, when the ant bites deep into the leaf vein, the “death grip,” because the ant is now locked to the leaf, providing a secure attachment for the fungus growing inside it. At this point, the ant is close to death, usually surviving for another 6 hours following its death grip. Its head is filled with fungal cells growing between muscle fibers, as well as around the brain and postpharyngeal gland[1]. Following the death grip, the ant’s mandible muscles atrophy, leaving its jaws locked into the leaf long after it’s dead.
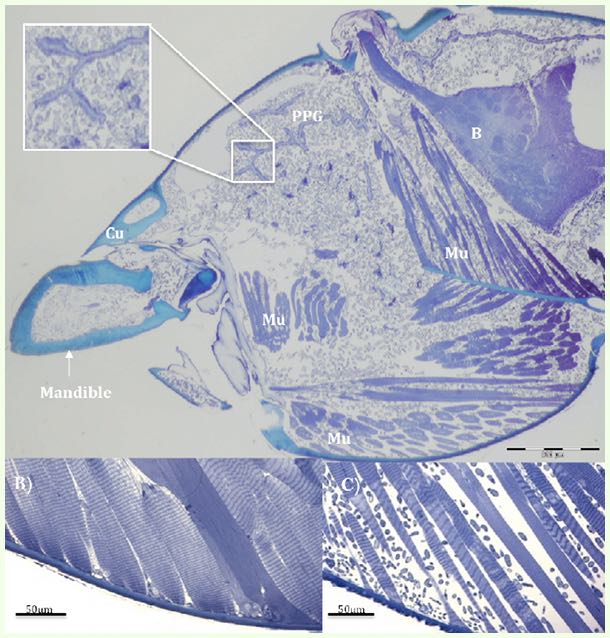
This image from the scientific paper shows a micrograph of an infected ant's head. It's the state of the ant's head when it bit down on the leaf, while it was still alive. Small grey blobs filling the head and mandible are the fungus. "PPG" is the postpharyngeal gland, "B" is the brain, "Mu" are muscles, and "Cu" is the cuticle (outer body of the ant). The small image in the lower left shows a close-up of what muscle would look like in a healthy ant. The small image in the lower right shows a close-up of the infected ant's muscle, right after it bit down on the leaf. The blobs between the muscle fibers are fungal cells.
About two to three days after the death grip, a fruiting body emerges from the dead ant’s head. It holds spores, which are released into the air, ready to be picked up by another tropical carpenter ant victim. Eventually, the ant’s remains fall to the ground. Scientists studying the understory where zombie ants are found attached under leaves have also found the remains of dead ants scattered on the ground – graveyards for past victims of the Ophiocordyceps fungus.
A paper in the journal BMC Ecology, published on May 9, 2011, describes the parasitism by a fungus on the tropical carpenter ant in Thailand. It’s a macabre description of how the ant is invaded by the fungus, which takes over its body, commands it to perform actions to ensure the survival and growth of the fungus, then uses its dead body to grow a stalk with spores that are ejected into the surrounding area, ready to infect other passing ants.
Back to post 1 A gland that secretes an odor unique to an ant colony.

A tropical carpenter ant killed by a parasitic fungus in Thailand. The ant had bitten into the leaf in a "death grip," and a fungal fruiting body with spores has emerged from its head. Image Credit: David P. Hughes, Penn State University.
|
 |
 |
 |
 |
 |
  a. a. | The line between fungus biology and late-night television just got blurrier.
A fungus that attacks living ants apparently manipulates their behavior for its own benefit, an international research team reports in the September American Naturalist.
When the Ophiocordyceps unilateralis fungus strikes, an infected ant climbs to a leaf not far off the ground (often on the north-northwest side of a tree), bites in and dies with jaws locked in place. Experiments now show that these low-hanging leaves give the fungus prime conditions for growing a spore-bearing spike out of the ant’s neck, says study coauthor David Hughes of Harvard University.
|
 |
 |
 |
 |
 |
  (1) (1) | Reproductive behavior and life cycle
As early as the 1940s it was reported that female wasps of this species sting a roach (specificially a Periplaneta americana, Periplaneta australasiae or Nauphoeta rhombifolia[1]) twice, delivering venom. A 2003 study[2] demonstrated using radioactive labeling that the wasp stings precisely into specific ganglia of the roach. It delivers an initial sting to a thoracic ganglion and injects venom to mildly and reversibly paralyze the front legs of the insect. This facilitates the second venomous sting at a carefully chosen spot in the roach's head ganglia (brain), in the section that controls the escape reflex. As a result of this sting, the roach will first groom extensively, and then become sluggish and fail to show normal escape responses.[3] In 2007 it was reported that the venom of the wasp blocks receptors for the neurotransmitter octopamine.[4]
The wasp proceeds to chew off half of each of the roach's antennae.[1] Researchers believe that the wasp chews off the antenna to replenish fluids or possibly to regulate the amount of venom because too much could kill and too little would let the victim recover before the larva has grown. The wasp, which is too small to carry the roach, then leads the victim to the wasp's burrow, by pulling one of the roach's antennae in a manner similar to a leash. Once they reach the burrow, the wasp lays a white egg, about 2 mm long, on the roach's abdomen. It then exits and proceeds to fill in the burrow entrance with pebbles, more to keep other predators out than to keep the roach in.
With its escape reflex disabled, the stung roach will simply rest in the burrow as the wasp's egg hatches after about three days. The hatched larva lives and feeds for 4–5 days on the roach, then chews its way into its abdomen and proceeds to live as an endoparasitoid. Over a period of eight days, the wasp larva consumes the roach's internal organs in an order which guarantees that the roach will stay alive, at least until the larva enters the pupal stage and forms a cocoon inside the roach's body. Eventually the fully-grown wasp emerges from the roach's body to begin its adult life. Development is faster in the warm season.
|
 |
 |
|
 |





